|
The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. He then made modifications to the structure developed by William Crookes in the 19th century. 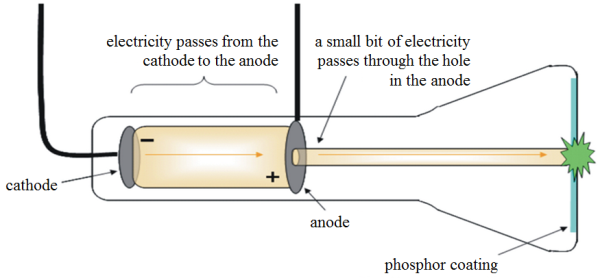 
Goldstein began his experiments with Crookes tubes during the decade of the 70s. When did Eugen Goldstein start experimenting with tubes? On the other hand, he is sometimes credited as the discoverer of the proton, which I observe in the vacuum tubes where he observed the cathode rays. Was there an atomic model of Goldstein? Goldstein did not propose an atomic model, although his discoveries allowed the development of the atomic model of Thomson. Is there an atomic model of Eugen Goldstein? One of his most outstanding legacies consisted in the discovery of what today are known as protons, together with channel rays, also known as anodic or positive rays. This discovery was made through experimentation with cathode ray tubes, in 1886. Goldstein identified the existence of protons as equal and opposite charges to electrons. How did Eugen Goldstein discover the existence of protons? His scientific work includes experiments with electrical phenomena in gases and cathode rays. What did Eugen Goldstein do for a living?Įugen goldstein He was a prominent German physicist, born in present-day Poland in 1850. We know them today as x-rays which are part of the electromagnetic spectrum. Roentgen discovered that if he directed these rays toward a paper plate coated with barium platinocyanide, the plate became fluorescent. In 1895, Wilhelm Roentgen, experimenting with cathode rays, discovered new and different kinds of rays. How did Wilhelm Roentgen contribute to the atomic theory? Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.”Įugen Goldstein married margarete fiedler and had 3 children. Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. 
The positive particle was named the proton. This resulted in The positive particle had a charge equal and opposite to the electron. 4 How did Eugen Goldstein discover the existence of protons?Ĭathode Ray Tube Eugene Goldstein discovered positive particles by using a tube filled with hydrogen gas (this tube was similar to Thomson’s tube).3 What did Eugen Goldstein do for a living?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
